Translate this page into:
Surgical emphysema following shoulder arthroscopy – A case report and review of a less recognized complication

-
Received: ,
Accepted: ,
How to cite this article: Sivaram R, Shanmugasundaram S, Thangavel M, Mohammed EA. Surgical emphysema following shoulder arthroscopy – A case report and review of a less recognized complication. J Arthrosc Surg Sports Med 2020;1(2):230-5.
Abstract
Arthroscopy is an established modality of treatment for various conditions of the shoulder attributable to its minimally invasive nature. The possibility of surgical procedure-related complications is usually low. However, there have been sporadic reports of surgical emphysema following shoulder arthroscopy that can range from simple subcutaneous emphysema to life-threatening pneumothorax (PT) and pneumomediastinum (PM). The mechanism of this phenomenon is still elusive. We report the case of a 41-year-old female without pre-existing lung problems who developed extensive subcutaneous emphysema, PT, and PM after shoulder arthroscopy in the absence of an identifiable tracheal injury and masquerading as angioedema. We have extensively reviewed available literature and the mechanisms proposed have been analyzed. Acknowledging the existence of such a complication along with high degree of suspicion will help tackle the problem early and avert unforeseen complications.
Keywords
Mediastinal emphysema
Pneumothorax
Arthroscopy
Shoulder
Subcutaneous emphysema
Angioedema
Emphysema
Shoulder arthroscopy
INTRODUCTION
Arthroscopy of the shoulder is a surgical procedure that is gaining lot of popularity because of its less invasive nature and because of low complication rates.[1,2] However, there appears to be an inclination for this surgery to be associated with surgical emphysema (SE), which can present as a simple subcutaneous emphysema (SCE) or as a complicated pneumomediastinum (PM) and/or pneumothorax (PT). Such complications can lead to lot of anxiety to the surgeon and the patient alike. We report a case of a simple arthroscopic procedure of the shoulder, complicated by PM and PT, masquerading as angioedema of the face. We also analyze available literature on this serious, but less recognized complication. Acknowledging this complication is necessary to keep the surgeon and patient informed.
CASE REPORT
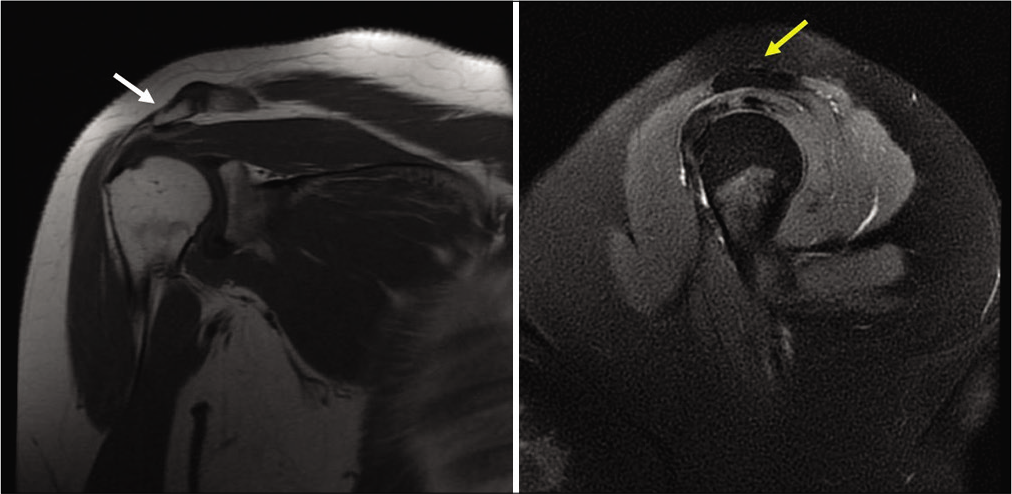
A 41-year-old female who was diabetic without any respiratory comorbidities and a body mass index of 30.1 presented with 3 months history of significant left shoulder pain that persisted despite physical therapy for 2 months. Her physical examination revealed shoulder impingement (Neer’s test and painful arc sign positive) with mild acromioclavicular joint (ACJ) tenderness. Her shoulder radiograph appeared normal [Figure 1]; magnetic resonance imaging revealed a type 2 and slanting acromion with supraspinatus tendinosis with 2 mm focal interstitial tear in distal fibers along with early degenerative changes in the ACJ [Figure 2]. Her pre-operative chest radiograph was normal [Figure 3]. She was taken up for arthroscopic acromioplasty and debridement of the ACJ under American Society of Anaesthesiology Grade 2, general anesthesia in beach chair position. She was induced with midazolam 2 cc, fentanyl 100 mcg, propofol 150 mg, and cisatracurium 10 mg and intubated with size 7.5 endotracheal tube (cuff inflated with 4–5 ml of air). She was ventilated in volume control mode with tidal volume 500 ml, positive endexpiratory pressure 5, respiratory rate 12/min, and 40% oxygen with 60% air. A diagnostic arthroscopy with a standard posterior portal revealed normal intra-articular structures. Subacromial arthroscopy revealed significant bursal tissue inflammation, without any bursal side supraspinatus tear. Acromioplasty was done using a 4.5 mm shaver blade, a 5 mm burr, and an electrocautery device attached to suction with a posterior viewing portal and a posterolateral instrumentation portal. An additional anteroinferior portal just lateral to the coracoid and 1.5 cm inferior to the ACJ was used to debride the ACJ. Throughout the procedure, the pump pressure was maintained at 50 mmHg and was kept at the same level as the operating table. The patient’s oxygen saturation was maintained at around 98% and the blood pressure at around 100/70 mmHg. No cannulae were used and portals were closed with 3-0 Ethilon. The intraoperative time was 1 h. Around 4 h postoperatively, the patient developed severe pain and uneasiness in the chest wall and operated site while still maintaining good saturation and vitals. After an injection of pethidine 50 mg, she developed swelling over the neck, chest, and left eyelid. Angioedema secondary to pethidine was suspected and injection hydrocortisone 100 mg was administered. Subsequently, crepitus extending from the chest wall to the left upper eyelid was noted. Bilateral air entry was equal and oxygen saturation was 91%. An anteroposterior radiograph of the chest showed evidence of SCE [Figure 4]. Computed tomography (CT) revealed PM with bilateral PT in addition with an intact trachea and bronchi [Figure 5]. The patient was managed conservatively with supportive oxygen, incentive spirometry, bronchodilators, and analgesics. Subsequent chest radiographs on the 2nd and 3rd post-operative days showed resolving SCE and PT. Her oxygen saturation improved to 98% and she was discharged on the 5th post-operative day. She continued to remain asymptomatic and on the last follow-up at 6 months postoperatively, her shoulder range of motion and power were full and she had resumed full function.
- Radiograph of the left shoulder showing normal appearance.
- Magnetic resonance imaging of the shoulder revealing a type 2 and slanting acromion with supraspinatus tendinosis (white arrow – slanting acromion; yellow arrow – type 2 acromion).
- Normal pre-operative anteroposterior chest radiograph (white arrow - slanting acromion; yellow arrow - type 2 acromion).
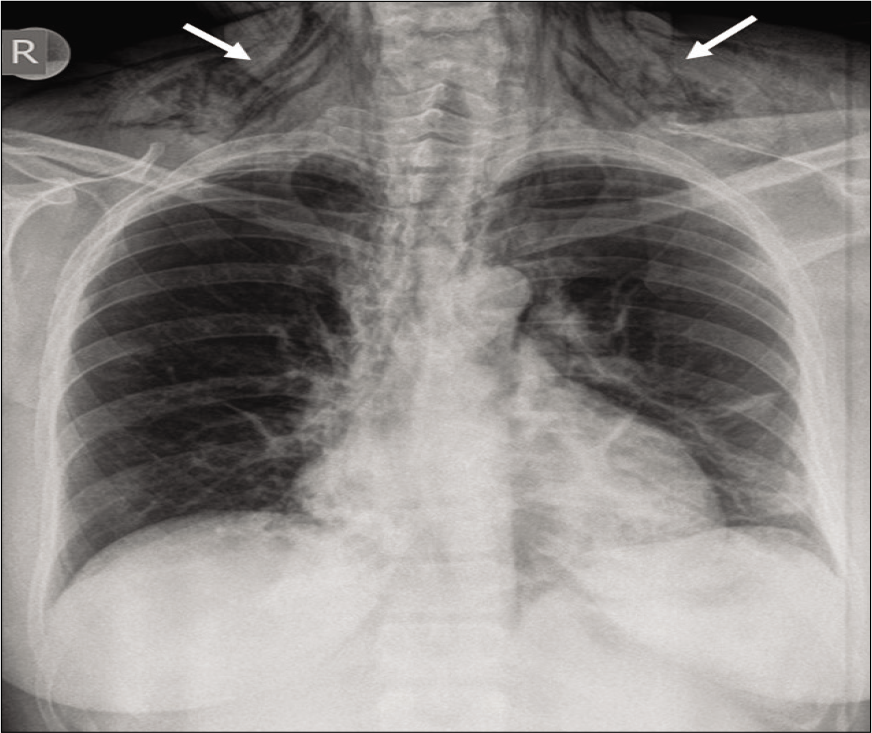
- Post-operative anteroposterior chest radiograph showing striations, a characteristic sign of subcutaneous emphysema (white arrows).
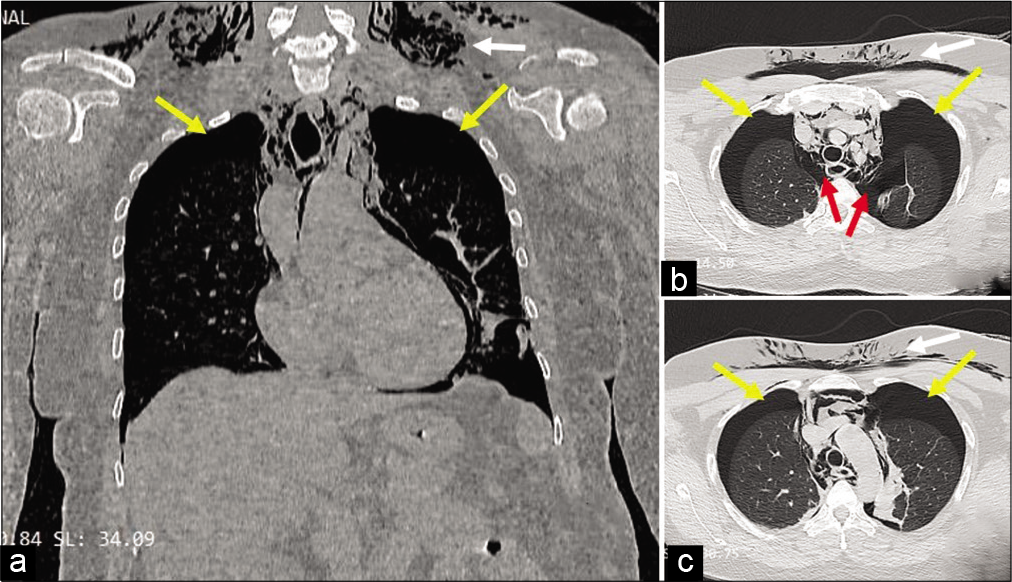
- Coronal section (a) and axial sections (b and c) of computed tomography showing subcutaneous emphysema (white arrows), bilateral pneumothorax (yellow arrows) and pneumomediastinum (red arrows).
DISCUSSION
SE is a state in which air permeates into the soft tissue under the skin.[3] A search on PubMed for “(emphysema AND shoulder arthroscopy)” and “(SE AND shoulder surgery)” yielded eight and 17 results in English language, respectively. The reports and their references were scrutinized. In all, there were 10 reports and 15 patients who had SE following shoulder arthroscopy. Among these, four underwent arthroscopic rotator cuff repair, four underwent arthroscopic subacromial decompression (SAD), one had shoulder arthroscopy followed by percutaneous Mumford procedure, two underwent debridement for glenoid labral tear with one having additional SAD, three underwent superior labrum anterior posterior repair with SAD, and one had shoulder arthroscopy without any mention of further details. A summary of the patient characteristics, surgical procedure, type of SE, and mechanism proposed is tabulated in [Table 1]. Five patients had SCE with PM, three had SCE with PT, two had SCE with both PT and PM, one had only extensive SCE, and four had PT without any mention of associated SCE or PM. Thirteen patients underwent general anesthesia with endotracheal intubation (ETI), of which two had additional scalene brachial plexus block (SBB), while two patients had only SBB.
| S. no. | Name of publication | Diagnosis | Surgical procedure | Complication | Diagnosis based on | Mechanism proposed |
|---|---|---|---|---|---|---|
| 1. | Lee et al., 1992 | Impingement syndrome | SAD (GA) | SE+PM+PT | CXR+CT | Intermittent shaving with suction causes drop in SA pressure sucking in air through portals, causing SE. Air extends through prevertebral fascia caused PM. Rise in mediastinal pressure secondary to PPV and expiration causes parietal pleura tear and PT |
| 2. | Lau et al.,1993 | Glenoid labral tear | Debridement (GA) | SE+PM | CXR+FOB | Loose junction between inflow tubing and plastic saline bag and multiple capsule puncture lead to extravasation of air and saline. Extension into prevertebral space through prevertebral fascia leads to PM |
| 3. | Dietzel et al.,., 1996 (4 patients) |
1. SLAP tear, synovitis 2. SLAP tear, impingement 3. Labral tear, impingement 4. Labral tear, impingement |
1. SLAP repair, synovectomy (GA) 2. SLAP repair, SAD 3. Labrum debridement, SAD 4. SLAP repair, SAD, rotator cuff imbrication |
PT | CXR | Smoking and pre-existing respiratory morbidity; – presence of bullae or bleb (pre-operative radiographs show only 20% of bullae) – rupture – alveolar rupture – PT, associated PPV in GA ; or due to increased intra-alveolar pressure during intubation; lateral decubitus position – more negative pleural pressure – high gradient from PPV in GA – rupture of Bleb |
| 4. | Calvisi et al.,., 2009 |
Subacromial impingement | Acromioplasty+ACJO (GA+SBB) | SE+PM | CXR+FOB+CT | (1) Inadvertent puncture of prevertebral fascia during scalene block leads to SE and PM, (2) Bernoulli effect results in increased outflow velocity leading to pressure drop in SA space and suction of air through portals; extension of air through the prevertebral fascia leads to PM |
| 5. | Knight et al., 2011 |
Rotator cuff tear | Cuff repair (GA with bougie) | SE+PT+PM | CXR+CT +FOB | Tracheal wall injury during intubation exacerbated by use of gum elastic bougie |
| 6. | Kamal et al., 2013 |
Impingement syndrome | Arthroscopy - SBB | SE+PT+PM | CXR+CT | (1) Possibly same as Lee et al.,. and Calvisi et al., (2) also possibly due to SBB, (3) possible traumatic PT during seizure episode postoperatively |
| 7. | Leander et al., 2016 |
SSC+SS tear | Cuff repair - GA+SBB | SE+PT | CXR+CT | (1) SBB leading to PT, (2) ET leading to PT by causing barotrauma in the presence of risk factors (smoking, lung disease), (3) use of shaver with suction/diathermy near pleural space, (4) thoracic trauma |
| 8. | Shariyati et al., 2017 |
Rotator cuff tear | Cuff repair under GA | SE+PT | CXR+CT | (1) Barotrauma caused by ET, (2) use of shaver and diathermy along with intermittent suction |
| 9. | Saseendar et al., 2014 | Severe AC joint arthrosis | P/C Mumford - GA | SE | CXR | Spiral flutes in power burr act as propeller in a contained space, pushes air into adjacent tissues, heat from burring expands air. SC tissue acts as one way valve, air gets sucked in further due to chest movements |
| 10. | Van et al., 2017 | Rotator cuff tear | Cuff repair - GA+SBB | SE+PM | CXR+CT | 1) GA+ET leading to tracheal tear, 2) rupture of pre-existing sub pleural blebs, 3) lower SA pressure/intraoperative loose junction between inflow valve and plastic bag leads to suction of air into closed compartment, air dissects into subcutaneous tissue and through prevertebral fascia causing PM, 4) early mobilization before portal site healing leads to suction of air into soft tissue with shoulder movements with soft tissue functioning as one way flap valve |
ET: Endotracheal intubation, GA: General anesthesia, SBB: Scalene brachial plexus block, SE: Subcutaneous emphysema, PT: Pneumothorax, PM: Pneumomediastinum, CXR: Chest X-ray, CT: Computed tomography, PPV: Positive pressure ventilation, ACJO: Acromioclavicular joint osteophytectomy, FOB: Fiber-optic bronchoscopy, GS: Gram stain, BC: Blood culture, SSC: Subscapularis, SS: Supraspinatus, P/C: Percutaneous, SAD: Subacromial decompression, SC: Subcutaneous
SCE, PM, and PT were previously unreported complication following shoulder arthroscopy with SAD until Lee et al. reported this in 1992.[4] Lau reported SCE and PM in 1993.[5] Causes of early post-operative SE after shoulder arthroscopy could be related to arthroscopy technique,[1] arthroscopic equipment,[1,3,6] general anesthesia (ETI and positive pressure ventilation [PPV]),[1,3,6,7] regional anesthesia (scalene block and associated pleural injury),[6,8] and patient-related factors (chronic airway disease, smoking, and anatomical abnormalities).[3,7] All patients in literature, except one,[5] underwent subacromial procedures.
Our patient was neither short-statured which may lead to difficult intubation nor did she have any pre-existing lung conditions or history of smoking which can predispose to spontaneous emphysema and PM secondary to rupture of pre-existing bullae.[3,7]
The possibility of developing SCE, PT, and PM following interscalene block[6] does not apply to our patient since no attempt was made to deliver regional anesthesia. Our patient underwent ETI with GA and PPV which are known to cause tracheal injury and subsequent PM, PT, and SCE.[4,7] Longer durations of PPV carry larger risk due to rupture of alveoli.[5] In addition, barotrauma due to increased airway forces and transpulmonic pressure gradient[6] is known to result in PT. However, our patient neither had any difficulty in intubation nor had any drop in saturation during the procedure, thus making ETI-related causes less likely. Moreover, patient’s postoperative CT did not reveal any tracheal tear till the main stem bronchus. We did not perform a fiber-optic bronchoscopy considering that her CT scan was normal and she showed progressive improvement in her oxygen saturation. CT scan is also the ideal investigation for diagnosis of PT.[6] Deitzel et al., in their series, quoted that higher negative pleural pressure on the operated side as compared to the non-operated side in a lateral position to be the reason behind PT. However, this mechanism does not apply to our patient as she was operated in a beach chair position.[7]
We postulate two mechanisms for the development of SE, PM, and PT in our patient. The first is, as suggested by Lee et al., the use of the arthroscopic shaver with an intermittent suction and without a working cannula causes a transient fall in the subacromial pressure in relation to the atmospheric pressure.[4,8] Furthermore, a Bernoulli effect could have induced a favorable state, thereby causing entry of air through the portals into the subacromial space[1,8] and, in turn, is pushed into the nearby tissues, leading to significant SE. This entrapped air permeates into the prevertebral space through the prevertebral fascia, which is part of the deep cervical fascia, leading onto a PM. Furthermore, increased mediastinal pressure with PPV and expiration causes rupture of the parietal pleura causing PT.
Injury to the parietal pleura is a well-known cause for PT. However, the portals used were far away from the established anatomical landmarks of the parietal pleura, and hence, an iatrogenic injury of the parietal pleura is very unlikely. Moreover, bilateral PT is unlikely in iatrogenic parietal pleural injury.
The second possible mechanism could be that the arthroscopic burr acts as a propeller in a contained space and tends to push air and fluid into the adjacent tissues, as described by Saseendar et al.[9] The heat generated by the bone burr leads to an expansion of air and aids in dissection through the subcutaneous tissue.
Postoperatively, our patient had left orbital swelling that mimicked angioedema, for which intravenous hydrocortisone was advised. However, angioedema was subsequently ruled out due to sparing of the lips and evidence of palpable crepitus in the neck and chest areas and radiographic confirmation of subcutaneous emphysema with its characteristic striations.[10] It is also pertinent to differentiate from SE caused by necrotizing fasciitis which advances rapidly and has a definitive infective focus and a higher death rate.[11] The presentation was on the same day after surgery, while gas-producing bacteria require around 18 h to form clinically significant gas[11] and there were no local signs of sepsis in the shoulder. Moreover, the laboratory risk indicators for necrotizing fasciitis, C-reactive protein, white cell counts, creatinine, sodium, and hemoglobin were normal and her chest radiograph and CT confirmed the diagnosis of SE.
CONCLUSION
This report not only throws light on a less recognized but potentially dangerous complication of shoulder arthroscopy that could initially mimic angioedema but also its various surgical and anesthetic causes. Subacromial procedures appear to be more predisposed to SE than intra-articular procedures. Knowledge of this complication and addressing it early can reduce morbidity and relieve anxiety for both the surgeon and the patient alike.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
Dr. Saseendar Shanmugasundaram is on the Editorial Board of the Journal.
References
- Massive emphysema and pneumothorax following shoulder arthroscopy under general anaesthesia: A case report. Arch Bone Joint Surg. 2017;5:459-63.
- [Google Scholar]
- Surgical emphysema of the neck following arthroscopic shoulder surgery. Br J Hosp Med. 2011;72:712-3.
- [CrossRef] [PubMed] [Google Scholar]
- Subcutaneous emphysema after shoulder arthroscopy. A case report and review of the literature. J Orthop. 2017;14:287-9.
- [CrossRef] [PubMed] [Google Scholar]
- Subcutaneous emphysema, pneumomediastinum, and potentially life-threatening tension pneumothorax. Pulmonary complications from arthroscopic shoulder decompression. Chest. 1992;101:1265-7.
- [CrossRef] [PubMed] [Google Scholar]
- Pneumomediastinum caused by subcutaneous emphysema in the shoulder. A rare complication of arthroscopy. Chest. 1993;103:1606-7.
- [CrossRef] [PubMed] [Google Scholar]
- Pneumothorax following shoulder arthroscopy under combined regional and general anaesthesia-a case report. Int J Surg Case Rep. 2016;24:73-6.
- [CrossRef] [PubMed] [Google Scholar]
- Spontaneous pneumothorax after shoulder arthroscopy: A report of four cases. Arthroscopy. 1996;12:99-102.
- [CrossRef] [Google Scholar]
- Subcutaneous emphysema and pneumomediastinum following shoulder arthroscopy with brachial plexus block: A case report and review of the literature. Arch Orthop Trauma Surg. 2009;129:349-52.
- [CrossRef] [PubMed] [Google Scholar]
- Extensive subcutaneous emphysema complicating a percutaneous Mumford procedure. Knee Surg Sports Traumatol Arthrosc. 2016;24:1907-10.
- [CrossRef] [PubMed] [Google Scholar]
- Subcutaneous emphysema mimicking angioedema. Indian Dermatol Online J. 2016;7:55-6.
- [CrossRef] [PubMed] [Google Scholar]
- Extensive subcutaneous emphysema resembling necrotizing fasciitis. Orthopedics. 2013;36:671-5.
- [CrossRef] [PubMed] [Google Scholar]






