Translate this page into:
Mid-term outcomes of arthroscopic osteochondral autograft transplantation for focal chondral defects of the knee

-
Received: ,
Accepted: ,
How to cite this article: Shekhar A, Reddy S, Patil S, Tapasvi S. Mid-term outcomes of arthroscopic osteochondral autograft transplantation for focal chondral defects of the knee. J Arthrosc Surg Sports Med 2021;2(1):41-6.
Abstract
Objectives:
Osteochondral autograft transplantation (OAT) for the management of cartilage defect involves direct transfer of hyaline cartilage to the defect site. The study aims to assess mid-term functional outcomes of arthroscopic treatment of focal chondral defects of the knee using the OAT technique.
Materials and Methods:
In this prospective follow-up study, patients who had previously undergone an OAT procedure for chondral defects of the knee at least 2 years prior were included. All the cartilage defects were treated arthroscopically with one or two plugs. Patients were followed up at 2 time points – in June 2016 and June, 2020. The International Knee Documentation Committee (IKDC) score and Tegner activity scale (TAS) were recorded preoperatively and at both follow-ups.
Results:
There were 20 patients with 21 focal cartilage defects, comprising 14 males and 6 females, with an average age of 30.9 ± 7 years and mean body mass index (BMI) of 27.2 ± 4 kg/m2. The first follow-up was after a mean 42.4 ± 12 months and the second after mean 90.4 ± 11.9 months post-surgery. The IKDC score improved significantly at both follow-ups compared to pre-operative scores (P < 0.001). At the first follow-up, the mean change in IKDC score was 25.3 ± 16.7 with 14 patients (70%) achieving minimal clinically important difference (MCID) but only 5 (25%) achieving substantial clinical benefit (SCB). At the second follow-up, 16 patients (80%) achieved MCID and 8 (40%) achieved SCB in IKDC score, with a mean change of 28.3 ± 12.6. The TAS showed statistically significant improvement at the first follow-up (P = 0.011), but not at the second follow-up (P = 0.052).
Conclusion:
OAT for focal cartilage defects of the knee is an excellent option as it provides clinically significant and incremental improvement in functional results in the mid-term. The results are not inferior even when a concomitant ligament or meniscus surgery is performed. Higher age or BMI does not have a negative impact on the outcomes.
Keywords
Knee
Arthroscopy
Articular cartilage
Hyaline
Osteochondral autograft transplantation
INTRODUCTION
The articular surface of the knee is comprised of hyaline cartilage, which is highly adapted to sustain and survive cyclical loads and high compression forces.[1] Cartilage is avascular and aneural and hence, spontaneous healing of chondral injuries is difficult. Focal cartilage defects which are not adequately treated early, progress to more diffuse breakdown, and progressive degeneration.[1] It has been reported that between 63 and 66% patients undergoing knee arthroscopy have some degree of articular cartilage damage and about 5–11% of these are of International Cartilage Repair Society (ICRS) Grades III and IV. Furthermore, a single chondral defect is present in only 4–7% of patients.[2-4] The techniques for surgical management of focal chondral defects can be either reparative (microfracture or subchondral drilling); restorative (osteochondral graft implantation); or regenerative (autologous chondrocyte implantation). Each of these techniques have their merits and limitations.[5] Local considerations for selecting a particular technique are the size and site of the defect. Patient factors include the age of the patient, sports activity participation, and cost implications. It is imperative to address any ligament, meniscus, or mal-alignment issues before or simultaneously with cartilage treatment.[6]
Smaller cartilage lesions have been extensively treated since almost 60 years using the marrow stimulation techniques of subchondral drilling and microfracture.[7,8] The resultant cartilage which develops at the defect site is fibrocartilage in nature and has inferior biomechanical properties. Osteochondral transfer surgery involves single or multiple, autograft or allograft plug(s) that are implanted at the defect site. It offers the distinct advantage of restoring hyaline cartilage as a single-stage procedure. The osteochondral autograft technique provides excellent osseous support to the overlying hyaline cartilage. This viable living structure helps achieve congruent repair of the articular defect in a single stage. Smaller focal defects may be treated with a single plug and are termed as osteochondral autograft transplantation (OAT). Larger defects may be treated using multiple small diameter plugs and are termed as mosaicplasty.[9,10] The OAT technique involving one or two plugs allows for an all arthroscopic technique, thereby reducing the morbidity of the procedure.
The critiques of the OAT procedure are that it is technically challenging to accurately match graft contour and size with the defect site and that there is donor-site morbidity. Initial attempts at OAT was using grafts from the patella and posterior femoral condyles, but these failed early due to mismatch of the congruency.[11,12] At present, grafts are harvested from the non-articular peripheral areas of the trochlea of medial or lateral femoral condyles above the sulcus terminalis, or from the intercondylar notch.[13] OAT is not very feasible for large size defects due to these potential problems.[14] Besides these, the interface between native and grafted cartilage gets filled up with fibrous tissue and fibrocartilage, leading to a lack of homogeneity of the articular surface.[14]
The purpose of this study is to report our experience and mid-term functional outcomes of arthroscopic treatment of focal chondral defects of the knee using the OAT technique.
MATERIALS AND METHODS
This is a prospective study of a cohort of patients who were operated by a single surgeon between January 2011 to March 2014 and surgical data were retrieved retrospectively. The Institutional Review Board approval was obtained before starting this study. Patients were identified based on the following inclusion criteria: (1) Arthroscopic OAT for cartilage defect in central weight-bearing area of either femoral condyle; (2) contralateral normal knee; and (3) completed 2 years follow-up. The exclusion criteria were: (1) Incomplete operative records; (2) any ligament knee injury other than anterior cruciate ligament (ACL); (3) revision knee surgery for any indication. Twenty-four patients who had undergone OAT procedure in this interval were eligible, but only 20 agreed to participate in the study. Informed written consent was obtained from all patients before recruitment, including consent to use retrospectively collected clinical, radiological, and surgery data.
Data extraction
The demographic data and details of surgery were retrieved from the hospital medical records department. All the patients had a pre-operative magnetic resonance imaging (MRI) scan to diagnose concomitant ligament or meniscus pathology. A weight bearing scanogram of both lower limbs was performed to diagnose any mal-alignment. All surgeries were performed under neuraxial anesthesia. The cartilage defects were graded using the ICRS classification.[15] The preand post-debridement size of the defects was documented using an arthroscopic ruler (Smith & Nephew Inc., Andover, MA). All OAT procedures were performed arthroscopically using either the OATS® (Arthrex, Naples, FL) or Mosaicplasty® (Smith & Nephew Inc., Andover, MA) systems as indicated by the individual graft size requirement. A single plug was used in all patients except one. This patient had a 13 × 10 mm defect size after debridement. Simultaneous reconstruction of the ACL, meniscus repair or excision, and high tibia osteotomy were performed, based on the merits of each case. The functional assessment was recorded as per the International Knee Documentation Committee (IKDC) form[16] and Tegner activity scale (TAS).[17]
Follow-up
All these patients were followed at 2 time points, the first was in June, 2016 when the last patient had completed 2 years of surgery and this is reported as the short-term (about 2–5 years post-surgery) follow-up. The second follow-up was in June 2020, that is, 4 years after the previous follow-up and is reported as the mid-term (about 6–9 years post-surgery) follow-up. All the patients were examined clinically and patient reported outcome measures were recorded using the IKDC form[16] and TAS.[17] The psychometric parameters of minimal clinically important difference (MCID) and substantial clinical benefit (SCB) for detecting changes in IKDC score were used. MCID is the smallest difference in score that patients deem as being beneficial, whereas SCB is the value that is perceived by the patients as having caused significant improvement in health.[18] The MCID of IKDC score for an OAT procedure has been reported as 17 ± 3.9, while SCB is when the score improves by 30 ± 6.9.[19]
Statistical analysis
Data analysis was performed using STATA software version 15.01 (StataCorp., Texas, USA). Qualitative data variables are expressed as frequency and percentage (%). Quantitative data variables are expressed as mean, standard deviation (±), median, and minimum-maximum. The continuous variables were analyzed using non-parametric tests. Wilcoxon Sign-rank test used to find the difference between pre-operative and post-operative IKDC scores and TAS. Spearman’s co-relation rank test was used to find the co-relation between IKDC with age, body mass index (BMI), and size of lesion. Mann–Whitney U-test was used to find any difference in IKDC score among sub-group of patients who underwent concomitant ACL or meniscus surgery and isolated cartilage surgery only. P < 0.05 was considered as statistically significant with 95% confidence interval.
RESULTS
There were twenty patients with 21 cartilage lesions in the final analysis with 14 males and six females. The left knee was affected in eight patients and right knee in 12. The mean age of the participants at the time of surgery was 30.9 years (19–42 ± 7). The mean BMI was 27.2 kg/m2 (21–36 ± 4). The intra-operative details are described in [Table 1]. There was a statistically significant increase in the area of the lesion from 1.13 cm2 (0.50–3.14 ± 0.69) before debridement to 2.10 cm2 (0.94–4.52 ± 1.06) after debridement (P < 0.001) [Figure 1]. Twelve patients had an associated pathology along with the cartilage lesion. The first follow-up was after a mean 42.4 months (26–66 ± 12) of surgery, while the second follow-up was performed at a mean 90.4 months (72–114 ± 11.9) after surgery. The changes in patient reported outcome measures are described in [Table 2].
| Etiology | Trauma | 19 |
|---|---|---|
| Osteonecrosis | 1 (Both MFC and LFC) | |
| Site of Chondral defect | Medial femoral condyle | 13 |
| Lateral femoral condyle | 8 | |
| ICRS Grade of defect[15] | 3A | 2 |
| 3B | 6 | |
| 3C | 6 | |
| 4A | 5 | |
| 4B | 2 | |
| Additional pathology | ACL Tear | 10 |
| Medial meniscus tear | 4 | |
| Lateral meniscus tear | 5 | |
| Both meniscus tear | 3 | |
| Varus malalignment | 1 |
MFC: Medial femoral condyle, LFC: Lateral femoral condyle, ACL: Anterior cruciate ligament, ICRS: International Cartilage Research Society
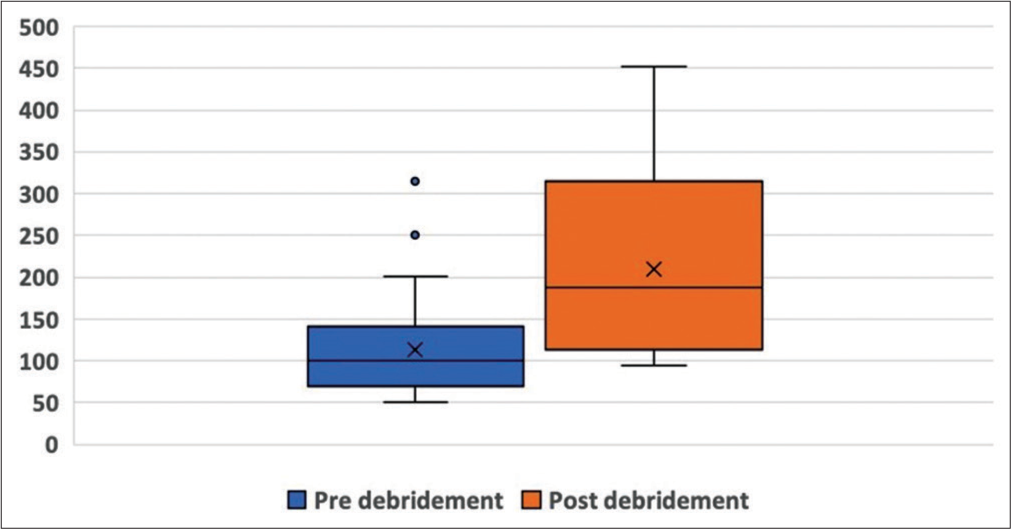
- Area of cartilage defect. A box and whisker plot of the area of the cartilage defects before (blue) and after (orange) debridement. The Y-axis represents area in mm2.
| Pre-operative | First follow-up (mean 42.4 months) | Second follow-up (mean 90.4 months) | |
|---|---|---|---|
| IKDC score | 56.3 (40.2–73.5±8.3) | 81.6 (34.5–100±14.8) | 84.6 (67.3–100±9.8) |
| P<0.001* | |||
| P<0.001* | |||
| Tegner activity scale | 4.3 (2–7±1.5) | 4.7 (3–8±1.3) | 4.3 (3–7±1.3) |
| P=0.011* | |||
| P=0.052* |
The IKDC scores were significantly improved at both follow-ups compared to pre-operative scores (P < 0.001) [Figure 2]. At the first follow-up, the mean change in IKDC score was 25.3 ± 16.7 with 14 patients (70%) achieving MCID but only 5 (25%) achieving SCB. At the second follow-up, 16 patients (80%) achieved MCID and 8 (40%) achieved SCB in IKDC score, with a mean change of 28.3 ± 12.6. The number of patients achieving MICD and SCB between the short-term and mid-term follow-up was calculated using two-tailed Fisher’s exact test and found to be statistically insignificant (P = 0.743). The TAS showed statistically significant improvement at the first follow-up (P = 0.011), but not at the second follow-up (P = 0.052). Spearman’s test coefficient showed poor co-relation of IKDC sores with age (r = –0.209, P = 0.214), BMI (r = –0.095, P = 0.688), and size of the cartilage defect (r = –0.471, P = 0.036) at final follow-up. The Mann–Whitney U-test found no association between IKDC score and performance of additional surgery at either short-term (P = 0.562), or mid-term (P = 0.164) follow-up. The distribution of IKDC scores and TAS based on performance of additional surgery(ies) is provided in [Table 3].
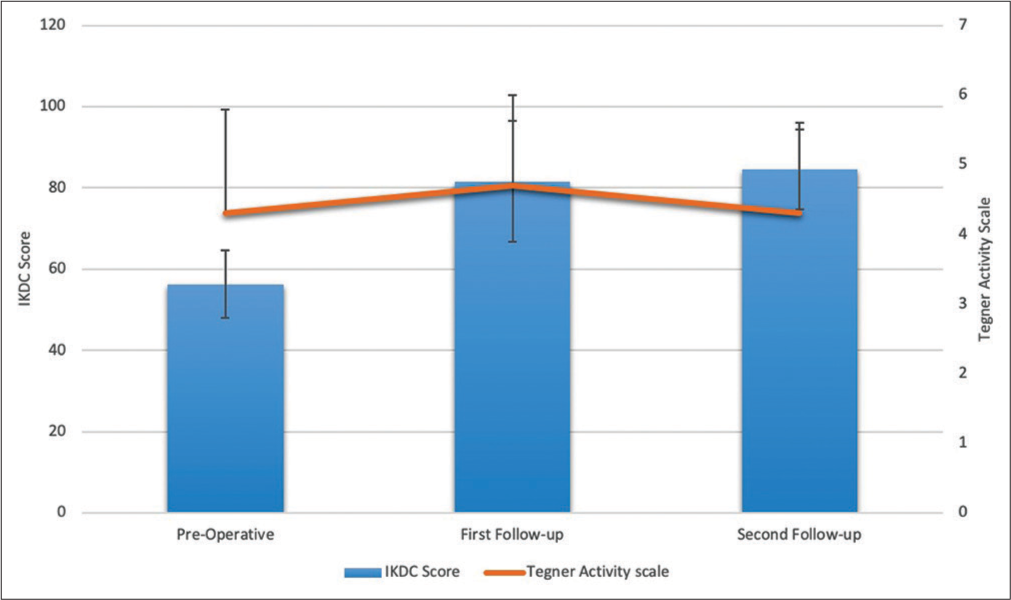
- Patient reported outcome measures. The bars depict mean International Knee Documentation Committee scores before surgery, at first and at second follow-ups. The mean Tegner activity scale is represented by the orange line at these 3 time points.
| Isolated OAT | OAT + ACL reconstruction | OAT + ACL reconstruction + meniscus surgery | OAT + meniscus surgery | |||||
|---|---|---|---|---|---|---|---|---|
| n=8 | n=6 | n=4 | n=2 | |||||
| IKDC | Tegner | IKDC | Tegner | IKDC | Tegner | IKDC | Tegner | |
| Pre-operative | 58.9±7.2 | 4.1±1.7 | 59.5±9.1 | 4.6±1.3 | 48.1±6.1 | 4±1.1 | 52.2±0.7 | 4.5±2.1 |
| 1stFollow-up | 85.2±8.4 | 4.5±1.6 | 79.1±11.1 | 5±1.2 | 87.1±12.3 | 4.7±0.9 | 63.8±41.4 | 4.5±2.1 |
| 2nd Follow-up | 88.6±6.7 | 4.1±1.3 | 80±11.5 | 4.6±1.2 | 87±9.5 | 4.2±1.5 | 77.8±14.9 | 4.5±2.1 |
OAT: Osteochondral autograft transplantation, ACL: Anterior cruciate ligament, IKDC: International Knee Documentation Committee, ±: Standard deviation
DISCUSSION
The most important finding of this study is that the functional outcomes measured using the IKDC score after arthroscopic OAT technique for focal chondral defects shows statistically significant improvements at short-term and this improvement persists in the mid-term. This improvement was clinically important in about three-fourth of patients although only about two-thirds achieved SCB. There was significant improvement in activity level using the Tegner scale seen in the short-term but this was not sustained at mid-term follow-up. It is plausible that the reduction in activity level over this time period could be due to age-related changes or lifestyle modification, not directly related to the knee function. Such reduction is knee function after an OAT procedure from 2 to 7 years has been reported previously as well.[20]
The short- and mid-term results of chondral defects treated with the osteochondral autograft transfer technique have been reported to be good in more than 85% patients.[21,22] In Hangody’s series of 789 femoral condyle defects treated using this technique, 92% patients had a good or very good outcome after a minimum 15 years follow-up.[23] In our series, 17/20 patients (85%) had a normal or nearly normal knee on objective IKDC scale at the mid-term follow-up. The results from our study support the use of OAT for the management of small chondral defects. Most patients achieved clinically relevant improvement in their functional outcome and this improvement is incremental over time. It has been reported by Chahla et al. that the MCID threshold is dynamic with progressively more patients achieving this with longer follow-up[24] and patients in our cohort also seem to follow this trend. We did not detect any specific variable in patients who did not achieve MCID of IKDC scores, except that their mean age (36.3 years) was higher than that of entire cohort. However, Spearman’s co-relation did not show any significant bearing of age on IKDC scores.
Small cartilage defects can be managed by either subchondral drilling or microfracture as well, which are simpler techniques with negligible immediate cost implications. This contrasts with OAT, which is a technically demanding procedure and entails additional instruments and cost. However, OAT allows for hyaline cartilage restoration and it is relatively simpler to match graft size and contour when only one or two plugs need to be implanted. The net benefits in terms of durability and incremental costs need to be factored in when comparing OAT with microfracture. Gudas reported significantly higher Tegner scores and significantly lower failure rate in athletes undergoing OAT than microfracture after 10-year follow-up. Athletes in the OAT group were more likely to achieve and maintain same level of sports participation in their cohort.[25] Solheim et al. have also reported superior outcome of OAT compared to microfracture at short-, mid-, and long-term for the treatment of 2–5 cm2 lesion in the distal femur.[26] None of the patients in our study had to undergo any revision procedure for failure of OAT, which implies 100% survival in the mid-term.
It is important that the correct technique is employed when performing OAT. Congruency of the graft surface with the surrounding tissue is critical and a 1 mm counter sink can afford protection in the initial period. If the graft surface protrudes above its surrounding, it may undergo necrosis due to excess stress concentration. Similarly, a graft counter sunk more than 2 mm may also fail due to lack of any surface stress that is necessary for the cartilage surface to thrive.[27] Care must be taken to ensure that the insertion is not forceful as multiple low force impactions are less damaging than a few forceful blows on the graft surface. It has been demonstrated that for an 8 mm graft, force threshold of 400 N (<10 MPa) is maintained so that the cell viability stays high.[28] The donor sites may be left empty and usually heal up with fibrous tissue. Donor site morbidity is rare but can cause post-operative pain.[29] No patient in this study specifically complained of pain or tenderness at the area of graft harvest at either follow-up.
Most of patients in this series (12/20) had an associated tear of the ACL and/or one or both menisci. If we are to consider the 19 patients with post-traumatic cartilage lesion, then the incidence of associated pathology is 63% in our cohort. The presence of knee instability has been an exclusion criterion in large prospective trials comparing OAT with either microfracture[25] or ACI.[30] It is possible that ACL or meniscus surgery performed in association with OAT for the cartilage lesion could confound the outcomes. However, the Mann–Whitney U-test failed to show any such association in our study. This is an encouraging indicator and implies that there is no negative impact on functional outcome of osteochondral transplantation when an ACL reconstruction or meniscus surgery is performed simultaneously. As a corollary, in a patient who primarily presents with instability or locking due to an ACL or meniscus tear and is detected to have a focal cartilage defect, the performance of an OAT procedure does not compromise the results of the ligament or meniscus surgery.
This study has several limitations. First, the number of participants in this study is small and hence the statistical analysis may be under-powered. Second, the surgical data collection was performed retrospectively from the available notes and relevant details of the surgery may have been missed. This would include details about the conformity of contour, graft recession/proudness, and status of the interface with normal cartilage. These variables are known to have a bearing on chondrocyte viability. Third, no follow-up MRI scan was performed to verify the healing of the osteochondral plugs or status of the implanted cartilage. Fourth, there is no comparative control group which would have allowed direct comparison of OAT with another technique. Finally, the associated injury and surgery performed in a majority of our patients could be a confounding factor, with unknown implications on the functional outcome.
CONCLUSION
OAT for focal cartilage defects of the knee is an excellent option as it provides clinically significant and incremental improvement in functional results in the mid-term. The results are not inferior even when a concomitant ligament or meniscus surgery is performed. Higher age or BMI does not have a negative impact on the outcomes.
Acknowledgment
The authors acknowledge the assistance received from Mr. Shrivallabh P. Sane (Lecturer in Biostatistics, Maharashtra Institute of Mental Health, Pune) for the statistical analysis.
Declaration of patient consent
Institutional Review Board permission obtained for the study.
Financial support and sponsorship
Dr. Tapasvi reports personal fees and non-financial support from SMITH AND NEPHEW, personal fees from ARTHREX, personal fees from STRYKER, personal fees from CONMED, personal fees from ZIMMER BIOMET, personal fees from JAPPEE MEDICAL PUBLISHERS, outside the submitted work. Other authors have nothing to declare.
Conflicts of interest
Dr. Sachin Tapasvi is on the Editorial Board of the Journal.
References
- Preservation of knee articular cartilage. Sports Med Arthrosc Rev. 2018;26:e23-30.
- [CrossRef] [PubMed] [Google Scholar]
- Cartilage injuries: A review of 31,516 knee arthroscopies. Arthroscopy. 1997;13:456-60.
- [CrossRef] [Google Scholar]
- Articular cartilage defects in 1, 000 knee arthroscopies. Arthroscopy. 2002;18:730-4.
- [CrossRef] [PubMed] [Google Scholar]
- Articular cartilage lesions in 993 consecutive knee arthroscopies. Am J Sports Med. 2004;32:211-5.
- [CrossRef] [PubMed] [Google Scholar]
- Surgical treatment of articular cartilage defects in the knee: Are we winning? Adv Orthop. 2012;2012:528423.
- [CrossRef] [PubMed] [Google Scholar]
- Management of chondral defects of the knee In: Hardikar SM, ed. Hardikar's Operations: Text and Atlas. New Delhi: Jaypee Brothers Medical Pvt. Ltd.; 2019. p. :91-105.
- [Google Scholar]
- A method of resurfacing osteoarthritic knee joints. J Bone Joint Surg. 1959;41:618-9.
- [Google Scholar]
- Microfracture: Surgical technique and rehabilitation to treat chondral defects. Clin Orthop Relat Res. 2001;391(Suppl 391):S362-9.
- [CrossRef] [PubMed] [Google Scholar]
- Arthroscopic autogenous osteochondral mosaicplasty for the treatment of femoral condylar articular defects. A preliminary report. Knee Surg Sports Traumatol Arthrosc. 1997;5:262-7.
- [CrossRef] [PubMed] [Google Scholar]
- Arthroscopic osteochondral autograft transplantation in anterior cruciate ligament reconstruction: A preliminary clinical study. Knee Surg Sports Traumatol Arthrosc. 1996;3:262-4.
- [CrossRef] [PubMed] [Google Scholar]
- The use of a lateral patellar autologous graft for the repair of a large osteochondral defect in the knee. J Bone Joint Surg Am. 1995;77:65-72.
- [CrossRef] [PubMed] [Google Scholar]
- The illustrative osteochondral cylinder transfer techniques for cartilage repair: The OATS technique. The Illustrative Book of Cartilage Repair 2020:123-35.
- [CrossRef] [Google Scholar]
- Current solutions for the treatment of chronic articular cartilage defects in the knee. EFORT Open Rev. 2020;5:156-63.
- [CrossRef] [PubMed] [Google Scholar]
- Development and validation of the international knee documentation committee subjective knee form. Am J Sports Med. 2001;29:600-13.
- [CrossRef] [PubMed] [Google Scholar]
- Rating systems in the evaluation of knee ligament injuries. Clin Orthop Relat Res. 1985;198:42-9.
- [CrossRef] [Google Scholar]
- Determining the clinical importance of treatment benefits for interventions for painful orthopedic conditions. J Orthop Surg Res. 2015;10:24.
- [CrossRef] [PubMed] [Google Scholar]
- Clinically meaningful improvement after treatment of cartilage defects of the knee with osteochondral grafts. Am J Sports Med. 2018;47:71-81.
- [CrossRef] [PubMed] [Google Scholar]
- Arthroscopic autologous osteochondral grafting for cartilage defects of the knee. Am J Sports Med. 2007;35:2014-21.
- [CrossRef] [PubMed] [Google Scholar]
- Autologous osteochondral grafting in the knee: Indication, results, and reflections. Clin Orthop Relat Res. 2002;401:170-84.
- [CrossRef] [PubMed] [Google Scholar]
- Arthroscopic autogenous osteochondral transplantation for treating knee cartilage defects: A 2-to 5-year follow-up study. Arthroscopy. 2004;20:681-90.
- [CrossRef] [Google Scholar]
- Autologous osteochondral grafting-technique and long-term results. Injury. 2008;39(Suppl 1):S32-9.
- [CrossRef] [PubMed] [Google Scholar]
- Defining the minimal clinically important difference and patient acceptable symptom state for microfracture of the knee: A psychometric analysis at short-term follow-up. Am J Sports Med. 2020;48:876-83.
- [CrossRef] [PubMed] [Google Scholar]
- Ten-year follow-up of a prospective, randomized clinical study of mosaic osteochondral autologous transplantation versus microfracture for the treatment of osteochondral defects in the knee joint of athletes. Am J Sports Med. 2012;40:2499-508.
- [CrossRef] [PubMed] [Google Scholar]
- Randomized Study of Long-term (15-17 years) outcome after microfracture versus mosaicplasty in knee articular cartilage defects. Am J Sports Med. 2017;46:826-31.
- [CrossRef] [PubMed] [Google Scholar]
- Effects of small incongruities in a sheep model of osteochondral autografting. Am J Sports Med. 2004;32:1842-8.
- [CrossRef] [PubMed] [Google Scholar]
- Effect of osteochondral graft insertion forces on chondrocyte viability. Am J Sports Med. 2008;36:1726-32.
- [CrossRef] [PubMed] [Google Scholar]
- Evaluation of donor site intrinsic healing response in autologous osteochondral grafting of the knee. Arthroscopy. 2002;18:95-8.
- [CrossRef] [PubMed] [Google Scholar]
- Autologous chondrocyte implantation and osteochondral cylinder transplantation in cartilage repair of the knee joint. A prospective, comparative trial. J Bone Joint Surg Am. 2003;85:185-92.
- [CrossRef] [PubMed] [Google Scholar]






